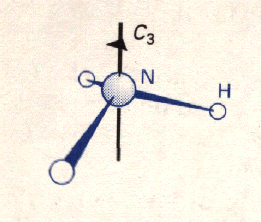
An NH3 Molecule
Though symmetry appears in almost every branch of science, it is perhaps most omnipresent in the field of chemistry. Indeed, a science which is so dependent on structure is a natural home for symmetry. More and more, symmetry has come to play an integral role in the deeper understanding of molecular structure and reactivity. And more and more, we realize that the fundamental building blocks of the world are not arbitrary clumps of matter, but highly symmetrical forms.
The primary classification of symmetry used by chemists is called the Schoenflies system, which deals with the discussion of individual molecules and is essentially the same as the grouping discussed in the mathematics section. (The Hermann-Mauguin system is used for crystals and crystallography.) However, there are a few changes in terminology. A symmetry operation is an action that leaves an object looking the same, e.g. a rotation (This is known as a transformation in mathematics). The symmetry operation is performed with respect to a symmetry element, e.g. an axis, plane, or point (Note: a symmetry element in chemistry is not to be confused with an element in a group). Just as mathematicians will try to identify all the symmetries of an object or pattern, chemists will try to identify all the symmetries of an individual molecule. The molecules are classified according to symmetry operations that leave a single common point unchanged; the set of these symmetry operations is a point group. For example, an NH3 molecule contatins the identity element, C3 rotational symmetry, and three planes of reflection - so it belongs to the point group C3v. For more information on symmetry notation click notation here.

An NH3 Molecule
Why is this useful? A molecule's structure can predict its characteristics. One trait that symmetry is commonly used to predict is polarity. A polar molecule is defined by having a permanent electric dipole moment - the charge distribution of the individual atoms doesn't even out. Any component of a dipole that exists in one direction is canceled by an opposing dipole. In the case of water, the perpendicular component of the dipole associated with the OH bond is canceled by the opposite dipole component of the second OH bond. However, its asymmetry leaves the parallel components uncanceled, resulting in a dipole moment parallel to its symmetry axis that runs straight down through the oxygen.
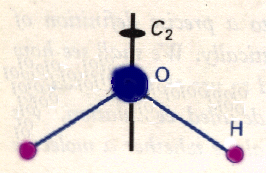
H2O
Another interesting trait related to symmetry is chirality. An object is said to be chiral when it cannot be superimposed on its mirror image, and whether or not a molecule is chiral determines whether it is optically active. The classic example of chirality is a pair of hands (hence scientists often refer to whether things are right-handed or left-handed). Rotations and translations are sometimes called proper symmetry operations because they transform a chiral object into itself. Reflection does not. Instead, reflection, an improper symmetry operation, converts chiral objects into their non-congruent mirror image. The chiral molecule and its mirror image are an enantiomeric pair.

Polarity and chirality are just two examples of symmetry applications in chemistry. Symmetry labels can also be given to molecular orbitals by using character tables, tables that characterize the different symmetry types possible in a point group. Character tables are derived from the formal language of group theory and are a good example of a quantitative use of symmetry. They can give us information about orbital degeneracy, as well as bonding. Crystallography and crystal symmetry are huge topics in science, for after all, crystals are essentially symmetrical lattice structures. Organic chemistry uses the spatial structure of molecules to predict reactions. The wave functions of electrons are also subject to symmetry considerations. .and this is just an introduction to symmetry applications in chemistry.
Symmetry,
Crystals, and Polyhedra
Crystallography 101
Symmetry Arguments in
Chemistry
The
Buckyball
Exercises in Point Group
Symmetry
Metric Tensors and Symmetry Operations in Crystallography
Page author: Sasie Sealy